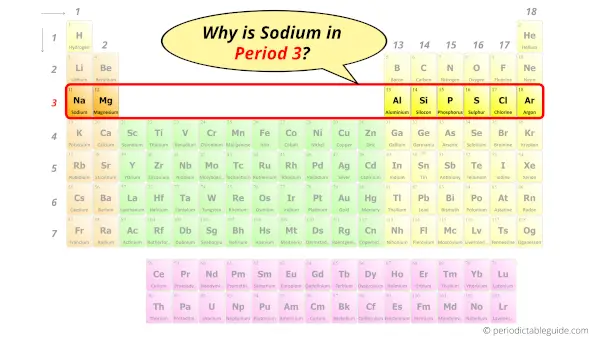
Sodium Element (Na) Information Appearance of Sodium So if you want to know anything about Sodium element, then this guide is for you. In fact, the table mentioned below is the perfect information box (Which gives you every single detail about the Sodium element in Periodic table.) Many organisms in the ocean depend on the proper concentration of ions in sea water to live.This is a SUPER easy guide on sodium element. Sodium ion in the form of sodium chloride is needed in the human body, but large amounts of it can cause problems, which is why one should not eat too much salt and other food items with huge sodium amount (such as biscuits with baking soda). Sodium is normally made by electrolysis of very hot sodium chloride that was melted. It is also found as sodium chloride in the earth's crust, where it is mined. It exists as an ion in chemical compounds. Sodium does not exist as an element in nature its easily removed valence electron is too reactive. Sodium compounds are used in soaps, toothpaste, baking and antacids. It is also used in the street lights that are orange, and ultra violet lights. It is used in the preparation of organic compounds. It is named after soda, a name for sodium hydroxide or sodium carbonate. He made it by the electrolysis of sodium hydroxide. Sodium was discovered by Sir Humphrey Davy, an English scientist, back in 1807.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
